Covalent Bonding
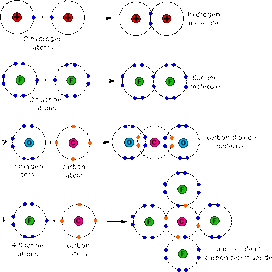
A covalent bond is a bond between two (or more) non-metals where they share a pair of electrons to gain a full valence shell.
A Covalent Structure:
A Covalent Structure:
- has low melting and boiling points
- has weak Van der Waal forces between molecules.This means the molecules can be easily separated, hence the low melting and boiling points.
